Bf3 dipole moment9/13/2023 
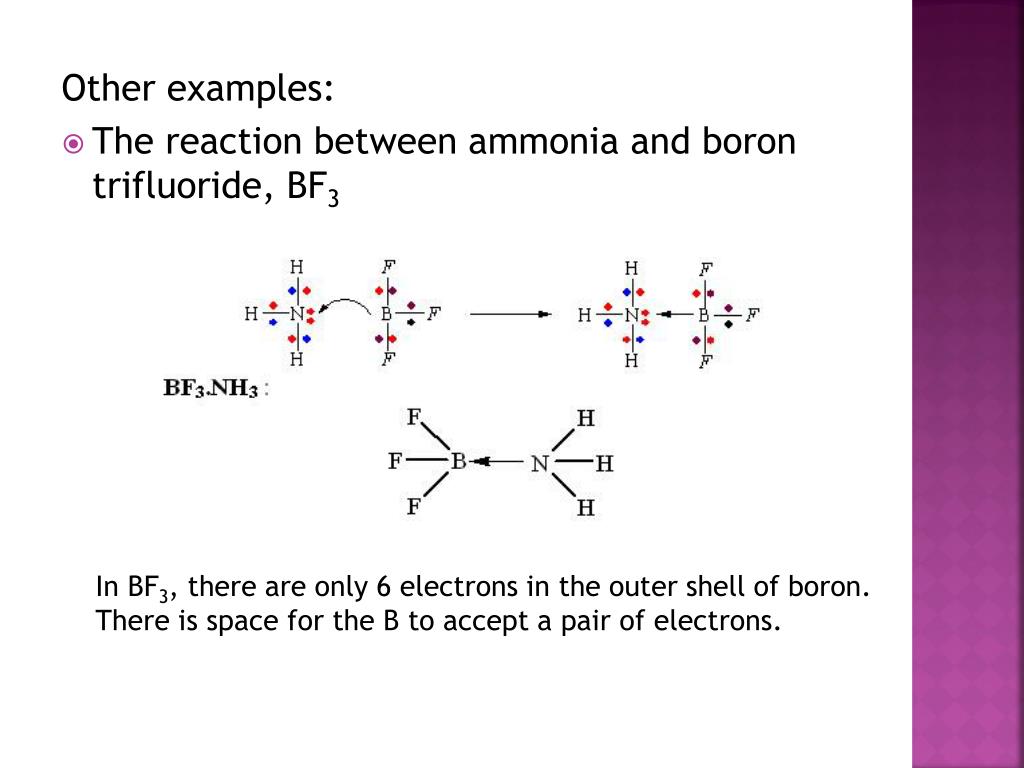
SiCl4 (silicon tetrachloride) is a nonpolar molecule. Check the full article “Is H2S polar or nonpolar?”. The difference in electronegativity values of the Hydrogen (2.2) and Sulfur (2.58) atoms makes H 2S a slightly polar molecule.įurthermore, the existence of two lone pairs on opposing sides of the two Hydrogen atoms makes the molecule more polar and results in the bending shape geometrical structure of H2S. Hydrogen sulfide (H 2S) is a colorless gas with a strong “rotten egg” odor. Is Hydrogen sulfide (H2S) polar or nonpolar? Even if each B-F bond is polar, the net dipole moment is zero because adding the bond vectors cancels everything out. Because the Boron-Fluorine bonds are all 120 degrees apart, any net dipole in that plane is canceled out. In BF3, the central boron atom has sp2 hybridized orbitals, resulting in an unfilled p orbital on the Bron atom and trigonal planar molecular geometry. Is BF3 Polar or Nonpolar?īF3 is a non-polar compound. This geometry forms an equilateral triangle with a 120-degree angle on each side. The structure formed in the plane shows that BF3’s molecular geometry is trigonal planar (central atoms are surrounded by three-terminal atoms). The primary distinction between ammonia and boron trifluoride is that ammonia is a polar molecule while boron trifluoride is a nonpolar molecule. What is the difference between ammonia and boron trifluoride?īoron trifluoride is an inorganic compound with the chemical formula BF3, whereas ammonia is an inorganic compound with the chemical formula NH3. If you have any questions please feel free to post a comment. Some of the frequent questions are given below. Related Links HCN Lewis Structure| Step By Step Construction N 2O Lewis Structure| Laughing Gas SiO2 Lewis Structure Co2 Polar or Nonpolar H2S Lewis Structure & Molecular Geometry SO2 Polar or Nonpolar Frequently Asked Questions
0 Comments
Leave a Reply.AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
